HKUST Researchers Unveil a Non-classical Nucleation Process That Enhances Ice Formation on Surfaces
Scientists from the Hong Kong University of Science and Technology (HKUST) have recently discovered a non-classical nucleation process that can greatly facilitate ice formation on foreign surfaces. This finding lays the foundation to predict and control crystallization processes.
Ice is omnipresent and profoundly impacts our daily life, influencing areas such as climate change, transportation, and energy consumption. Understanding the process of ice formation can decelerate the rate at which glaciers melt and sea levels rise and alleviate other major environmental concerns. Since ice formation is mainly governed by ice nucleation followed by the growth of the nuclei, scientists have put in a great effort to understand the thermodynamics and kinetics behind the nucleation processes. Ice nucleation can occur in two distinctive ways: homogeneously in bulk water or heterogeneously on the surface of a solid material, where heterogeneous ice nucleation (HIN) is the predominant mode of ice formation on earth. However, unlike homogeneous ice nucleation, the water-surface interactions present in HIN make the nucleation process sensitive to surface properties. Understanding how surfaces impact the nucleation process is a promising approach to better predict and control crystallization processes.
A common model used to quantify nucleation kinetics based on a thermodynamic framework, classical nucleation theory (CNT), suggests that water molecules must form an ice nucleus of critical size before a crystallization process occurs. The formation of the critical ice nucleus is associated with a single free energy barrier, which needs to be overcome to trigger further ice growth. However, over the years, both experiments and simulations have revealed that CNT is often insufficient to describe some complex nucleation processes. Consequently, CNT has been a subject of immense debate, and non-classical nucleation theories have been alternatively proposed.
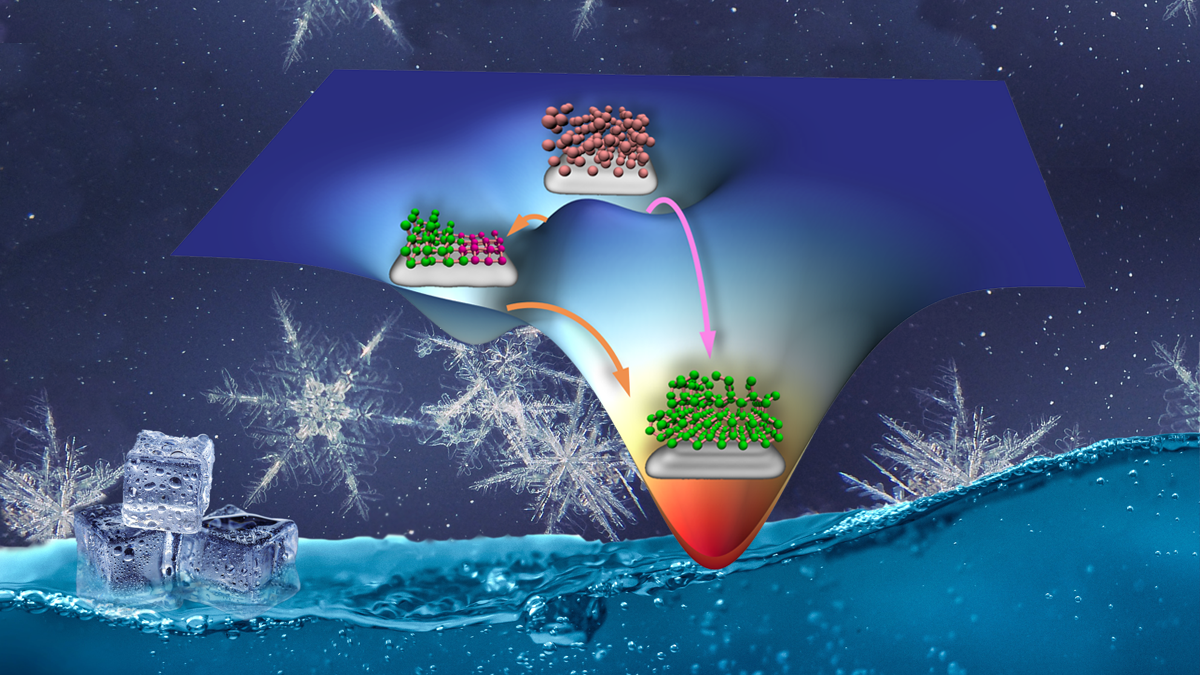
Different from CNT, which is based on overcoming a single free energy barrier, non-classical nucleation theories suggest that nucleation processes consist of two or more steps separated by multiple free energy barriers. Although non-classical nucleation theories may be a more sustainable model, the atomistic mechanisms and structural evolutions during nucleus formation in non-classical nucleation pathways are not well known; and remains a challenge for experimental techniques to unravel.
Now, for the first time, a group of researchers at HKUST led by Prof. Xuhui Huang from the Department of Chemistry combined Markov State Models (MSMs) – which model long-timescale dynamics of chemical molecules - and transition path theory (TPT) – which describes the reaction pathway of rare events - to elucidate the ensemble pathways of HIN. MSMs identify intermediate states of disordered ice mixtures and compare parallel pathways (classical vs. non-classical). This advantage helped unravel the underlying mechanisms of non-classical nucleation processes and the co-existence of the two pathways.
These researchers show that the disordered mixing of ice stabilises the critical nucleus and makes the non-classical nucleation pathway as accessible as the classical pathway, whose critical nucleus mainly consists of potential energy-favoured ice. They also discovered that at elevated temperatures, the nucleation process prefers to proceed via the classical pathway since the potential energy contributions, which favour the classical pathway, prevail.
“Not only does our work uncover the mechanisms of non-classical nucleation processes, but it also demonstrates how the combination of MSMs and TPT offers a powerful framework to study structural evolutions of ice nucleation processes,” said Prof. Huang. “More importantly, this method can be extended to other crystal nucleation processes that are challenging to study, which will open new doors for scientists attempting to predict and control crystallization processes.”
The findings were recently published in the scientific journal Nature Communications. The first author of this work: Dr. Chu Li is a long-time HKUST affiliate who completed his PhD, and currently conducts his post-doctoral training at HKUST.